De Chancourtois plotted the atomic weights on the surface of a cylinder with a circumference of 16 units, corresponding to the approximate atomic weight of oxygen. de Chancourtois proposed a classification of the elements based on the new values of atomic weights given by Stanislao Cannizzaro’s system of 1858. Get a Britannica Premium subscription and gain access to exclusive content.Īttempts were later made to show that the atomic weights of the elements could be expressed by an arithmetic function, and in 1862 A.-E.-B. In fact, no general agreement had been reached among chemists as to the classification of elements for nearly half a century after the systems of classification of compounds had become established in general use.
:max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
Relationships were discerned more readily among the compounds than among the elements it thus occurred that the classification of elements lagged many years behind that of compounds.

This rapid expansion of chemical knowledge soon necessitated classification, for on the classification of chemical knowledge are based not only the systematized literature of chemistry but also the laboratory arts by which chemistry is passed on as a living science from one generation of chemists to another. The early years of the 19th century witnessed a rapid development in analytical chemistry-the art of distinguishing different chemical substances-and the consequent building up of a vast body of knowledge of the chemical and physical properties of both elements and compounds. © American Chemical Society ( A Britannica Publishing Partner) See all videos for this article
Printable periodic table chemistry how to#
Printable periodic table chemistry series#
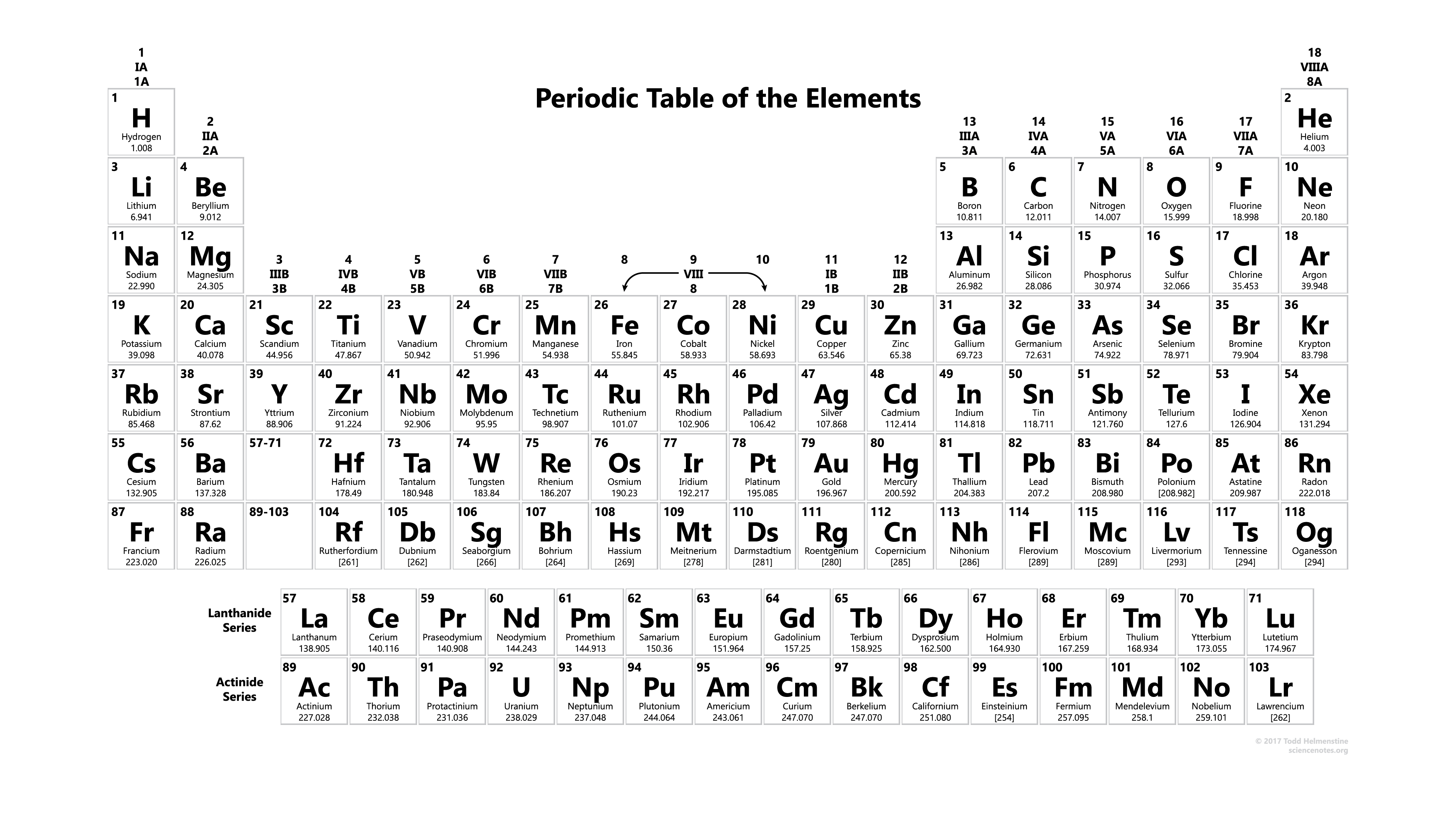
S-block – entails helium, hydrogen, alkali metals, and alkaline earth metals.į-block – normally placed below the table, this block presents the lanthanides and actinides series of metallic elements.Ĭells – these important components of a periodic table can entail various pieces of information, including the element’s atomic number and the one or two-letter atomic symbol, representing the element’s Latin name e.g “No” for “Nobelium.” Other information may include: Additionally, there are four blocks, each containing different groups Nevertheless, elements with identical chemical characteristics are assigned a group name and, sometimes, given the same color. Columns and rows are known as “Groups” and “Periods”, respectively. It is important to note that periodic tables are split into cells with each cell representing an element.
